Registry

Nairobi Cancer Registry was established in the year 2001 after consultations between the National Cancer Institute (NCI) – Office of International Affairs, International Agency for Research on Cancer (IARC), Ministry of Health (MOH) and Kenya Medical Research Institute (KEMRI). It is situated at the Centre for Clinical Research (CCR), KEMRI headquarters, Nairobi.
In 2009, recognising the problems of recording large numbers of cases with limited staff, an INCTR budget was established to support case finding for Nairobi residents only. The registry was a founder member of the East African Registry Network (in January 2011), and further development is on its way.
 The registry has attempted to register all cancer cases reported in various health institutions in Nairobi. Initially, the methodology was to recruit cancer registrars from employees of medical records service including hospitals, oncology clinics, labs and death registers, laboratory and clinical services. Pathology laboratories have not been employed as primary source because the demographic information availability is limited. These part time Medical Records Officers were trained on basic cancer registration techniques by the registry to collect data on all newly diagnosed cancer cases from the year 2000 in their respective hospitals, laboratories and radiotherapy units. The registry pays them some allowances as per cases abstracted on monthly basis. The data were submitted to the registry supervisor in KEMRI for verification and subsequent entry into a computer. HIV status has been included on the data collection forms. The CanReg4 software provided by IARC has been used. There is currently provision for 3 full time contractual registrars, and for part time data collectors in 4 major hospitals.
The registry has attempted to register all cancer cases reported in various health institutions in Nairobi. Initially, the methodology was to recruit cancer registrars from employees of medical records service including hospitals, oncology clinics, labs and death registers, laboratory and clinical services. Pathology laboratories have not been employed as primary source because the demographic information availability is limited. These part time Medical Records Officers were trained on basic cancer registration techniques by the registry to collect data on all newly diagnosed cancer cases from the year 2000 in their respective hospitals, laboratories and radiotherapy units. The registry pays them some allowances as per cases abstracted on monthly basis. The data were submitted to the registry supervisor in KEMRI for verification and subsequent entry into a computer. HIV status has been included on the data collection forms. The CanReg4 software provided by IARC has been used. There is currently provision for 3 full time contractual registrars, and for part time data collectors in 4 major hospitals.
Since its inception, data collection has included all cases, irrespective of place of residence, diagnosed in the hospitals in the registration area. The amount of data to be processed is vast. Potential workload is high. In 2006 for example, data on more than 3000 cases were entered into the computer (of which rather more than 60% were Nairobi residents).
The data items collected are relatively standard. They include:
1. Patient Details: First name, given/maiden name, last name (surname), ID number, age/date of birth, gender, concurrent illness, current residence, place (district) of birth, religion and tribe/ethnicity.
2. Tumour: Incidence date, basis of diagnosis, primary site/topography (ICDO) code, histology/morphology (ICD-O) code, behaviour, grade and stage at diagnosis (TNM).
3. Treatment(s): Initial and subsequent treatments – surgery, radiotherapy, chemotherapy, hormone therapy & symptomatic.
4. Sources of data: Hospital/laboratory name, hospital number, laboratory report number, date of abstraction
5. Follow-up: Patient status (alive or dead) as at date of incidence, last date of contact with physician/health-care provider or if dead - date of death and cause of death, hospice number.
Treatment modality is recorded as Y/N. Since 2009, as part of the agreement with INCTR, HIV status has been included on the data collection forms (it can be found relatively easily in records from KNH, where testing on admission is routine). However, HIV data have not to date been entered on CANREG (The current system makes no allowance for this variable).
Sources of information
The following health institutions currently submit their data to NCR:
i). Government and Private Hospitals
Registrars collect data from Medical Records Service areas. They use disease index cards and patient-care service registers to identify cancer cases in both in-patient and out-patient departments. However a few hospitals have established computer based disease indices which are also used to identify cancer cases.
ii). Medical Laboratories
Most of the said government and private hospitals have various specialty laboratories including histology, haematology and cytology. NCR staff visit laboratories that are likely sources of cancer incidence data.
iii). Radiotherapy Treatment Centres
The radiotherapy units at Kenyatta National Hospital and The Nairobi Hospital - MITC attend to both old and new cancer patients who visit these facilities for radiotherapy services. NCR registrars regularly visit these units and carry out active case abstraction.
iv). The Nairobi Hospice
The Nairobi Hospice submits data to NCR and in most cases, these are patients referred from KNH and other health-care facilities within and outside Nairobi. The hospice gives up-to-date information on patients’ status. This is important for follow up and case assessment purposes.
v). Vital Statistics - Registrar of Births & Deaths (Death Certificates Office)
In 2006 NCR established a link with Vital Statistics, to access cancer-specific mortality data from the Registrar of Births & Deaths Office.
Registration area
 Population of the city of Nairobi was estimated at 3,138,369 at the 2009 census. Although the city is a large cosmopolitan centre located in the Central highlands predominantly occupied by Bantus, its population is made up of most of the ethnic groups in the country. Unlike the other provincial cities and towns in Kenya, the population of the city of Nairobi is a fairly good representation of the general population of Kenya. The city of Nairobi enjoys the status of a full administrative County/Region. There are 8 main administrative divisions (Central, Dagoretti, Embakasi, Kasarani, Kibera, Makadara, Pumwani and Westlands).
Population of the city of Nairobi was estimated at 3,138,369 at the 2009 census. Although the city is a large cosmopolitan centre located in the Central highlands predominantly occupied by Bantus, its population is made up of most of the ethnic groups in the country. Unlike the other provincial cities and towns in Kenya, the population of the city of Nairobi is a fairly good representation of the general population of Kenya. The city of Nairobi enjoys the status of a full administrative County/Region. There are 8 main administrative divisions (Central, Dagoretti, Embakasi, Kasarani, Kibera, Makadara, Pumwani and Westlands).
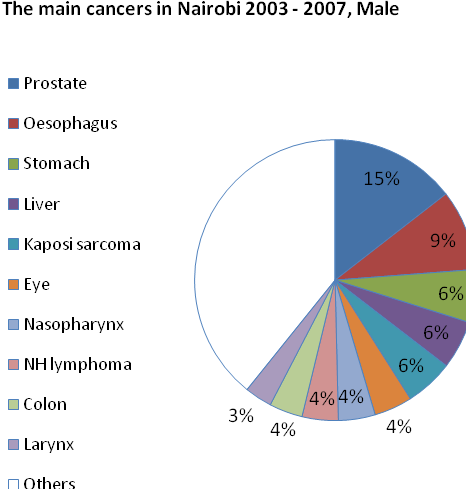
Results

Publications
Korir A, Okerosi N, Ronoh V, Mutuma G, Parkin M. Incidence of cancer in Nairobi, Kenya (2004-2008). Int J Cancer. 2015 Nov 1;137(9):2053-9. doi: 10.1002/ijc.29674. PubMed PMID: 26139540. Available for download below.
The registry has produced another comprehensive report on a four years activity 2004-2008. Available for download below.
The registry has produced a comprehensive report on the first 3 years activity 2000-2002 (published October 2006).
